A surgical site infection (SSI) is a post-surgical infection that can affect either the incision or deep tissue at the operation site. They have negative impacts on patients and hospitals, increasing the average patient’s hospital stay by approximately 6.5 days1. In Australia, hospital acquired surgical site infections may be associated with an average of $42,102 in extra costs2. Leukomed® by Leukoplast® is a range of dressings and other solutions to help to minimize the risk of SSIs.
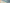
Safeguard your patient’s surgery
Leukoplast helps to protect your surgical results from surgical site infections

We help you minimize the risks of SSIs
Leukomed® by Leukoplast® is our solution to support the prevention of SSIs. A range of post-surgical dressings for use in multiple post-operative situations. We also offer a range of frameworks, guidelines, and support tools to assist you in minimizing the risk of SSIs, as well as ongoing access to our dedicated representatives.
Proven results with Leukomed® Sorbact®
The surface of Sorbact® irreversibly binds bacteria through a physical mode of action and it is safely removed4,5. This leads to an effective reduction of the bacterial burden in critically colonized or locally infected wounds6. Development of bacterial or fungal resistance is not expected. Clinical evidence shows that Leukomed® Sorbact® delivers results.
- 65% relative risk reduction of acquiring an SSI7
- 57% cost reduction when treating caesarian sections8
Discover our range of surgical site dressings
All Leukomed® surgical site dressings
Contact us
If you would like to know more about our dressings or simply want to discuss how we can support you in reducing the rate of SSIs in your facility, just fill out the form below and one of our dedicated representatives will be in touch shortly.
1 Global guidelines for the prevention of surgical site infection, second edition ISBN 978-92-4-155047-5 © World Health Organization 2018
2 Australian Commission on Safety and Quality in Health Care, March 2018, https://www.safetyandquality.gov.au/sites/default/files/migrated/Healthcare-associated-infection-detailed-fact-sheet.pdf
3 Getting it Right First Time, SSI National Survey, April 2019, https://gettingitrightfirsttime.co.uk/wp-content/uploads/2017/08/SSI-Report-GIRFT-APRIL19e-FINAL.pdf)
4 Husmark J, et al. Antimicrobial effects of bacterial binding to a dialkylcarbamoyl chloride-coated wound dressing: an in vitro study. J Wound Care. 2022 Jul 2;31(7):560-570.]
5 Ljungh, et al. Using the principle of hydrophobic interaction to bind and remove wound bacteria, Journal of Wound Care, Vol 15, No 4, 2006.
6 Mosti G, et al. Comparative study of two antimicrobial dressings in infected leg ulcers: a pilot study. J Wound Care. 2015;24(3):121-127.
7 Stanirowski PJ, et al. (2016b) Randomized controlled trial evaluating dialkylcarbomyl chloride impregnated dressings for the prevention of surgical site infections in adult women undergoing caesarean section. Surg Infect (Larchmt), 17(4): 427-35, 2016.
8 Stanirowski PJ, et al. Cost-effectiveness of a bacterial-binding dressing to prevent surgical site infection following caesarean section. J Wound Care. 2019 Apr 2;28(4):222-228.
9 Rousseau T, et al. An advanced transparent hydropolymer wound dressing for undisturbed post-op management of surgical wounds following hip and knee replacement: A prospective observational series; Int Wound J. 2021;1–7







